Amendments to the Tobacco Act entered into force on 1 February 2026
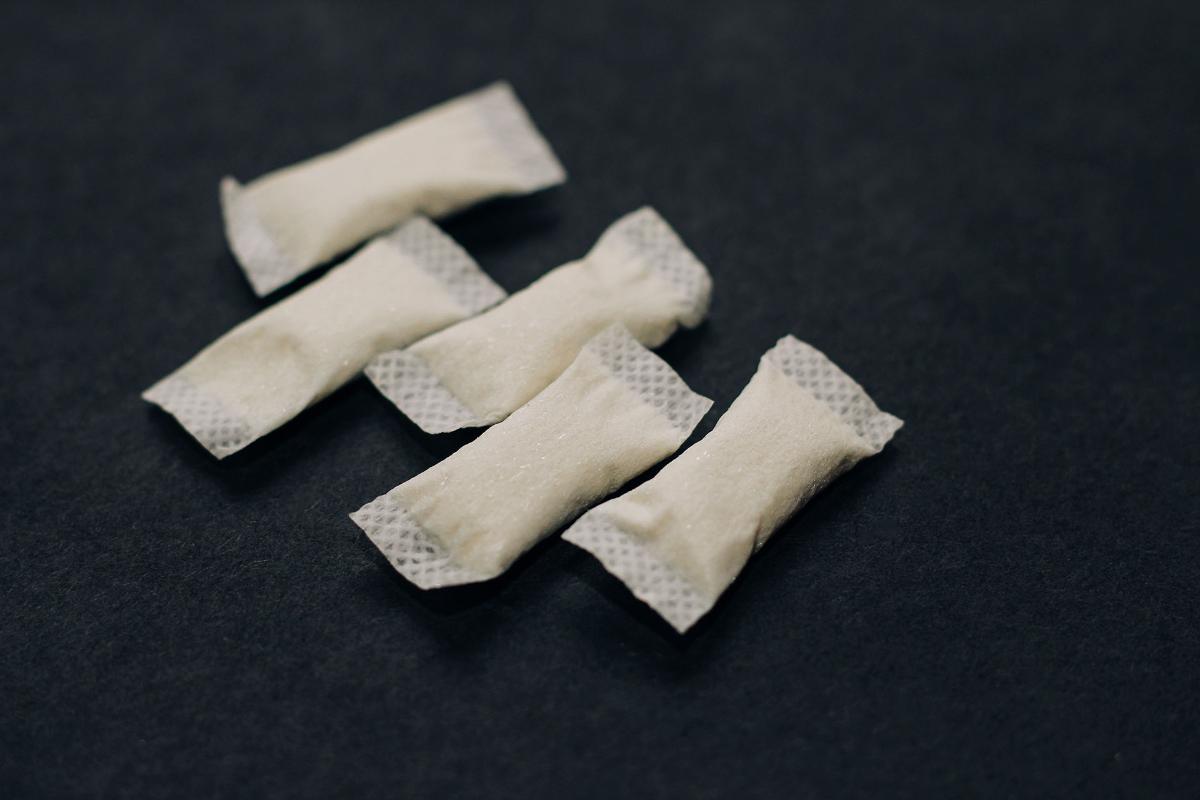
An amendment to the Tobacco Act concerning smokeless tobacco products, such as nicotine pouches, entered into force on 1 August 2025. The transitional period for the entry into force of certain amendments has now ended, and these amendments entered into force on 1 February 2026.
From 1 February 2026, the product notification obligation also applies to smokeless nicotine products. Manufacturers and importers of smokeless nicotine products are obliged to submit product notifications on products placed on the Finnish market. Product notifications must be submitted to the Finnish Supervisory Agency through the EU Common Entry Gate (EU-CEG) system. Read more about product notifications for smokeless nicotine products (especially nicotine pouches) in the Finnish Supervisory Agency’s instructions (in Finnish).
In addition to submitting a product notification, the manufacturer or importer of a smokeless nicotine product must pay the fees charged by the Finnish Supervisory Agency for the product in accordance with the Tobacco Act. Read more about the fees on our website (in Finnish).
Mandatory labelling on retail packaging
The requirements for retail packaging also entered into force on 1 February 2026. Now retail packaging of smokeless nicotine products must also be labelled as follows:
- a list of ingredients the products contains in descending order by weight;
- an indication of the product’s nicotine content and dosage;
- the manufacturer’s batch number;
- a recommendation to keep the product out of the reach of children;
- health warnings in Finnish and Swedish.
More stringent provisions on product properties
More stringent provisions were set for the properties of smokeless nicotine products.
- Maximum nicotine content limit: A smokeless nicotine product containing more than 16.6 milligrams of nicotine per gram may not be sold or otherwise handed over to the consumer.
- Restrictions on flavours: A smokeless nicotine product containing a characteristic smell or taste may not be sold or otherwise handed over to the consumer. However, a smokeless nicotine product may taste like tobacco, menthol, or mint.
- Product size restriction: Smokeless nicotine products may not be sold or otherwise handed over to the consumer in the form of ready-made units weighing less than 0.5 g or more than 1 g.
Prohibited additives and properties
The new Act contains more detailed provisions on prohibited additives and properties in smokeless nicotine products. A smokeless nicotine product may not be sold or otherwise handed over to the consumer:
- if it contains additives that are liable to create the impression that the product has a health benefit or presents reduced health risks compared to other similar products;
- if it contains stimulant compounds or other additives that are liable to create an impression of energy and vitality;
- if its additives have carcinogenic, mutagenic or reprotoxic (CMR) properties in its smokeless form;
- if it has flavourings in any of its components that allow the modification of the smell, taste, or smoke intensity of the product concerned;
- if it contains additives in quantities that increase the toxic or addictive effect, or the CMR properties of a tobacco product to a significant or measurable degree at the time of consumption.
The manufacturer and importer of smokeless nicotine products are responsible for ensuring that the product intended for sale or other supply in business operations complies with the relevant provisions and regulations. There must be no smokeless nicotine products on the Finnish market that do not meet the legal requirements.
Read more
Enquiries
Reija Kauppi, Head of Group, tel. +358 295255985